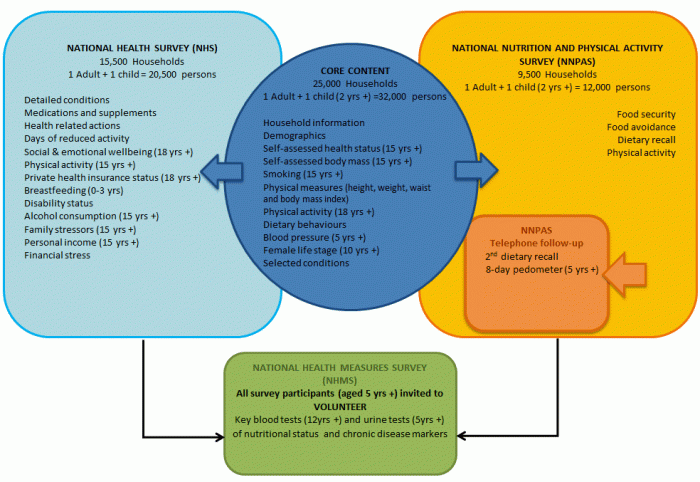
Results in this publication contain information from the National Health Measures Survey (NHMS), the biomedical component of the 2011–13 Australian Health Survey (AHS). Around 11,000 respondents aged 5 years and over across Australia voluntarily provided blood and/or urine samples, which were tested for a range of chronic disease and nutrition biomarkers. This publication is the first release of information from the NHMS and focuses on the test results for chronic diseases, including:
- Diabetes
- Cardiovascular disease
- Chronic kidney disease
- Liver function
Refer to Appendix A for the full list of tests conducted.
Diabetes
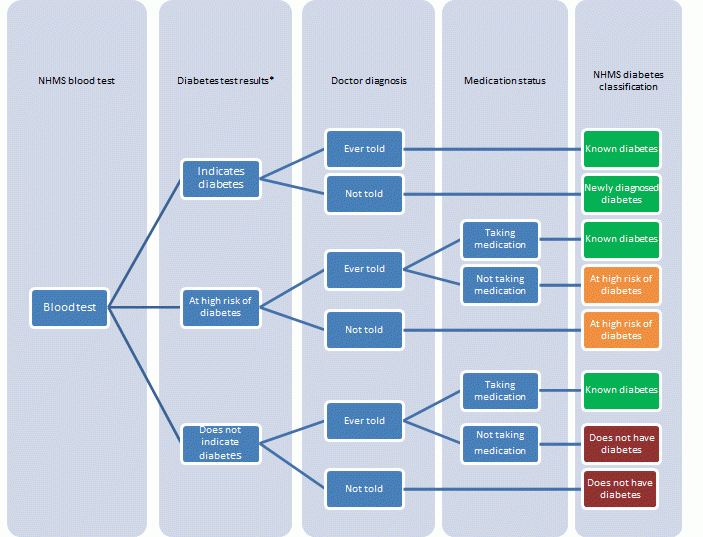
- In 2011–12, 5.1% of people aged 18 years and over had diabetes.
- This comprised 4.2% with known diabetes and 0.9% with diabetes newly diagnosed by the blood test results. This suggests that there was approximately one newly diagnosed case of diabetes for every four diagnosed cases.
- Men were more likely than women to have diabetes (6.3% compared with 3.9%). This was the case for both known diabetes and newly diagnosed diabetes.
- A further 3.1% of Australian adults were identified by their test results to be at high risk of diabetes.
Cardiovascular disease
- Around one in three Australian adults (32.8%) had high levels of total cholesterol according to their blood test results, yet only 10.1% of this group self-reported high cholesterol as a current health condition.
- One in three Australian adults (33.2%) had high levels of LDL 'bad' cholesterol and 23.1% had lower than normal levels of HDL 'good' cholesterol.
- In 2011–12, 13.9% of people aged 18 years and over had high triglycerides.
- Three in every four adults (76.4%) aged 45 years and over had dyslipidaemia. That is, they were taking cholesterol-lowering medication or had one or more of high total cholesterol, low HDL cholesterol, high LDL cholesterol or high triglyceride levels based on their test results.
Chronic kidney disease
- In 2011–12, one in ten (10.0%) Australian adults had test results that showed signs of chronic kidney disease, with similar rates for men and women.
- Around 4% of all adults were in Stage 1, 2.5% were in Stage 2 and less than 1% were in Stages 4–5.
Liver function
A range of factors, including fatty liver disease, infections and excessive alcohol consumption can prevent the liver from functioning properly. The NHMS included two tests for liver function: gamma glutamyl transferase (GGT) and alanine aminotransferase (ALT). These tests check the liver’s health and can detect liver damage.
- In 2011–12, 11.0% of Australian adults had abnormal levels of ALT in their blood, with men more likely to have the condition than women (13.8% compared with 8.3%).
- Around 2.1 million (or 12.4%) people aged 18 years and over were estimated to have abnormal levels of GGT.
Exposure to tobacco smoke
- In 2011–12, the pattern for cotinine exposure was very similar to that for self-reported smoking for most age groups.
- 87.0% of current smokers aged 18 years and over had cotinine levels indicating exposure to tobacco smoke, compared with only 5.7% of those who were ex-smokers and 0.3% of those who had never smoked.
Anaemia
Anaemia is caused from a decrease in either the number of red blood cells in the body or the quantity of haemoglobin within red blood cells. When a person is anaemic, their heart has to work harder to ensure that muscles and organs get the oxygen they need.
- In 2011–12, 4.5% of people aged 18 years and over had haemoglobin levels indicating a risk of anaemia, with women more likely to be at risk than men (6.4% compared with 2.5%).